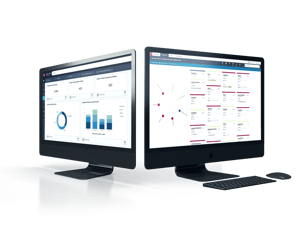
Organizing Brownfield Data Across Multiple Plants.

Pharma & Medical Device
Accelerate Insight. Assure Compliance.
Kobai unifies your clinical, manufacturing, and regulatory data into one connected knowledge layer — giving Pharma and Medical Device organizations the context they need to innovate faster and meet global compliance demands with confidence.
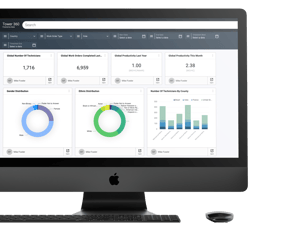
Critical Data Challenges in Pharmaceutical Manufacturing
Challenge 1
Multi-System Regulatory Compliance
Pharmaceutical manufacturers typically manage compliance data across numerous systems such as LIMS for laboratory data, MES for manufacturing execution, QMS for quality management, ERP for batch records, and document management systems for SOPs and validation protocols. Meeting FDA 21 CFR Part 11, EU Annex 11, and GMP requirements often requires demonstrating complete data lineage and audit trails across these disconnected systems.
Organizations report that creating comprehensive audit packages for regulatory submissions can require significant manual effort to trace data origins, transformations, and approvals across systems. Incomplete lineage documentation introduces compliance risk during regulatory inspections.
Challenge 2
Clinical Trial to Manufacturing Data Continuity
The path from clinical trials to commercial manufacturing involves data from clinical data management systems (CDMS), manufacturing pilot plants, scale-up facilities, and commercial production lines. Each phase may use different systems, data models, and identifiers for the same drug substance or formulation.
Industry practitioners note that reconciling formulation data, process parameters, and quality specifications across these phases can be challenging, particularly when modifications occur during scale-up. Gaps in data continuity may delay regulatory filings or complicate responses to manufacturing deviations.
Challenge 3
Supplier and Supply Chain Traceability
Pharmaceutical supply chains often span multiple tiers with critical raw materials, excipients, and API suppliers requiring stringent qualification and monitoring. Supplier data may exist in procurement systems, quality databases, regulatory tracking tools, and risk management platforms, each potentially using different supplier identifiers and classification schemes.
Getting a unified view of supplier quality history, audit findings, change notifications, and regulatory status can require manual data gathering across systems. Industry research suggests that supply chain disruptions increasingly create operational challenges, making rapid supplier evaluation critical.
Challenge 4
Batch Genealogy and Deviation Investigation
When quality deviations occur, investigators need to trace batch genealogy such as which raw materials from which suppliers went into which intermediates, combined in which equipment, under which process parameters, tested by which methods, and distributed to which customers. This information typically spans ERP, MES, LIMS, warehouse management, and distribution systems.
Practitioners report that deviation investigations can require substantial time to manually piece together complete batch histories from disconnected systems. Delayed investigations extend the time to resolve quality issues and resume production.
Challenge 5
Real-World Evidence and Post-Market Surveillance
Connecting clinical trial data, real-world evidence from EHRs, adverse event reports, manufacturing batch data, and distribution records requires integrating highly diverse data sources. Organizations pursuing precision medicine or investigating safety signals need to link molecular data, patient outcomes, manufacturing process data, and product distribution information.
The lack of semantic connections between these domains can limit the ability to quickly identify correlations between manufacturing variations and clinical outcomes, or to rapidly respond to emerging safety signals.
How Kobai Addresses These Challenges
Unify Patient Data for Proactive Insights
Connect siloed data from EMRs, labs, and IoT devices to create a 360-degree patient view. Identify at-risk patients and optimize care pathways.
Which patients with Type 2 Diabetes are showing high-risk vitals from their remote monitors and have a history of non-compliance with medication?

Explore the Use Case with Our Team
Please fill out the form below and we'll get back to you to understand your business case and explore a solution together.
Experience Our Product!
Looking to see our product in action? Please provide your details to request a personalized demo and discover how Kobai can help your business.Explore the Platform Behind the Possibilities
Dive deeper into the technology that powers connected data, graph intelligence, and enterprise-scale insights. Discover how Kobai’s platform is transforming how industries turn knowledge into action.